Its atomic number is 52, which is also the number of electrons we need to fit in the configuration. One might surmise that the failure of this structure to form complete octets must mean that this bond should be ionic instead of covalent. Remember the number of electrons hosted in each shell: s\rightarrow 2 s 2 p\rightarrow 6 p 6 d \rightarrow 10 d 10 f \rightarrow 14 f 14 Let's try with a heavier element, tellurium. electron diffraction by a number of authors and it has been shown that the short-range order of its atoms is different from that of the crystalline antimony. Calcium (Ca) Bromine (Br) Nitrogen (N) Antimony (Sb) Rubidium (Rb) Iron (Fe). Hydrogen atoms can naturally only have only 2 electrons in their outermost shell (their version of an octet), and as such there are no spare electrons to form a double bond with boron. How many electrons are in each of the following shells/subshells. Elements with 3 valence electrons are used for p-type doping, 5-valued elements for n-doping. 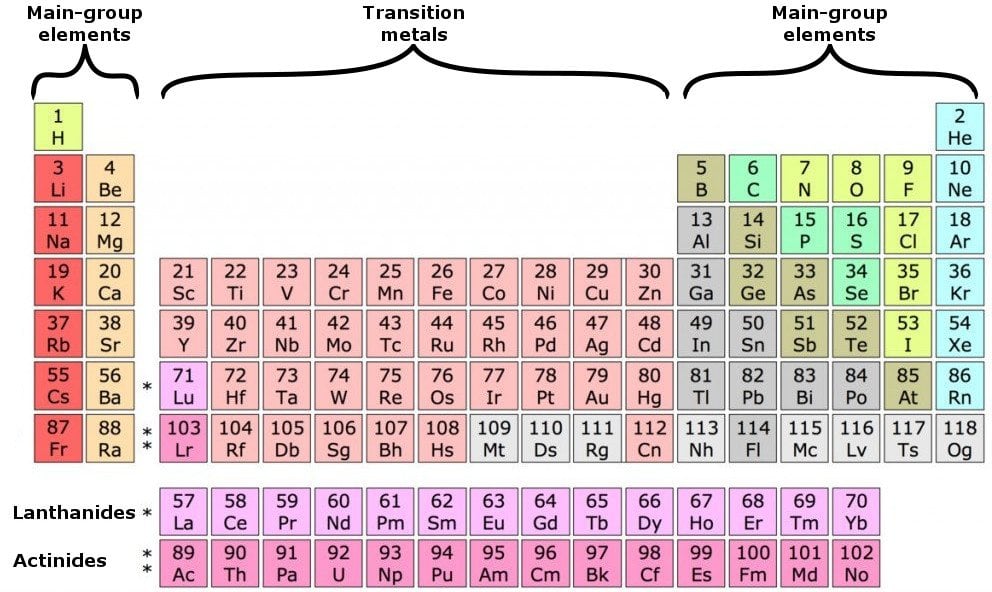
The dopant is integrated into the lattice structure of the semiconductor crystal, the number of outer electrons define the type of doping. The problem with this structure is that boron has an incomplete octet it only has six electrons around it. Other materials are aluminum, indium (3-valent) and arsenic, antimony (5-valent). \): The structure of \(BH_3\) is square planer.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
